A fluorescence microscope can recognize a wide range of fluorescent dyes, with wavelengths ranging from ultraviolet to infrared light. It’s more about how capable the instrument is of recognizing these dyes, based on its:
- Filter system
- Light source wavelength range
- Detector wavelength range
This means that, as long as the fluorescent dye corresponds with the microscope’s wavelength range, it will work.
Classification of Common Fluorescent Dyes
1. Classical Organic Dyes
The “old guard” of biology labs, these are standard for cell assays and immunofluorescence (IF), typically categorized by color:
| Fluorescence Category | Common Fluorophores | Features |
| Blue Fluorescence (UV Excitation) | DAPI, Hoechst 33342/33258 | Most common nuclear dyes; specifically bind to DNA |
| Green Fluorescence (Blue-Light Excitation) | FITC (Fluorescein) | Commonly used for immunofluorescence labeling |
| Alexa Fluor 488 | More stable and brighter than FITC; current industry benchmark | |
| Red / Orange Fluorescence (Green-Light Excitation) | TRITC, Texas Red | Classical red fluorophores |
| Cy3, Alexa Fluor 555 / 568 / 594 | Commonly used for multiplex staining | |
| Far-Red Fluorescence (Red-Light Excitation) | Cy5, Cy5.5, Cy7, Alexa Fluor 647 / 700 / 750 | Difficult to see with the naked eye, but easily captured by digital cameras; commonly used to avoid background autofluorescence |
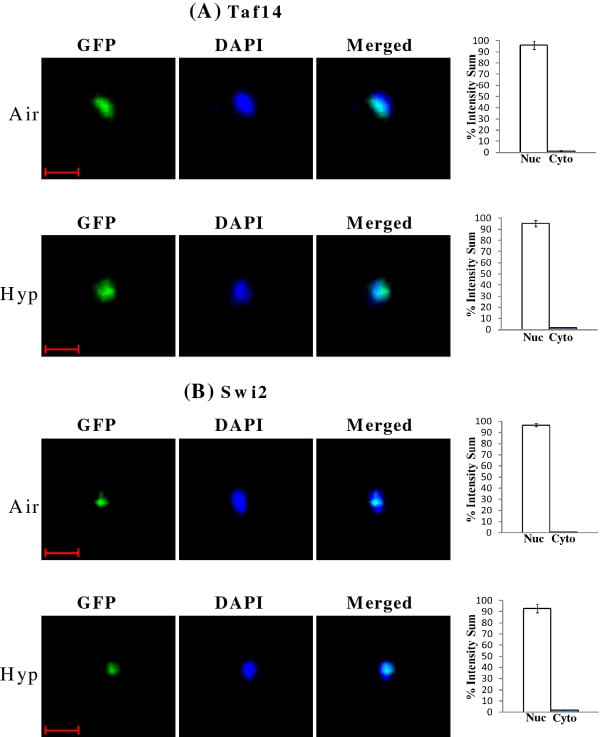
2. Fluorescent Proteins (FPs)
Proteins that have been genetically engineered to be expressed inside living cells without any additional staining:
- GFP (green fluorescence protein): Variants include EGFP.
- YFP/CFP/RFP (yellow/cyan/red fluorescence protein).
- mCherry/tdTomato: Highly sought-after red fluorescent proteins.
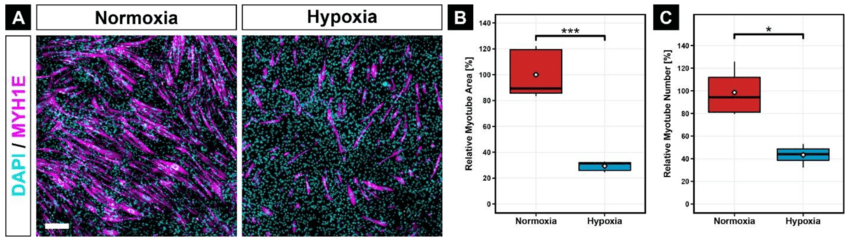
3. Special Functional Dyes
Used to monitor physiological states rather than just structure:
- Cell Viability: Calcein-AM (live cells), PI (propidium iodide, dead cells).
- Ion Indicators: Fluo-4 / Fura-2 (calcium concentration).
- Organelle-Specific: MitoTracker (mitochondria), LysoTracker (lysosomes).
4. Novel Fluorescent Materials
Quantum Dots: Extremely broad excitation, narrow emission, and high photostability.
Upconversion Nanoparticles (UCNPs): Emit visible light via NIR excitation.
Decisive Factor: Does the Fluorescence Microscope Hardware Support It?
When it comes to biological imaging, selecting the appropriate fluorescent dye is just the beginning. The critical factor that ensures clear visualization of the image is the compatibility of the imaging equipment and the fluorescent characteristics.
Even when choosing the proper dye, a substandard imaging system can produce an image that is completely dark or highly contaminated with noise.
The ultimate difficulty lies in distinguishing true fluorescence signals from background noise with extremely low signal levels.
- With 6 filter slots available, one can comfortably accommodate DAPI + GFP + RFP combinations while reserving extra slots for Cy5 and Cy7 filter cubes.
- When the filter slots are limited to 4 in number, there will hardly be any extra room left for far-red combinations after the configuration of normal three-color imaging.
Please refer to the label on the filter turret of the microscope. It will usually be labeled as U/B/G (DAPI/GFP/RFP). For Cy5/Cy7 imaging, in addition to placing the necessary filters, you also need to check if the sensitivity curve of the camera includes the range between 700nm and 850nm.
1. How Many Filter Cube Positions Does the Fluorescence Microscope Have?
The microscope’s capability depends not only on what dyes it can “see,” but also on how its optical path system is configured. Because: Number of filter cubes = Number of supported fluorescence channels
| Number of Filter Positions | Meaning |
| 3 positions | Basic three-color imaging |
| 5 positions | Expandable multi-dye imaging |
| 7+ positions | Advanced research / multiplex fluorescence imaging |
The total number of turret slots will depend directly on the type of microscope and positioning of the machine (clinic versus research).
- Basic/entry-level inverted microscopes: come equipped with 3 to 4 slots, DAPI blue, GFP green, RFP red, and one brightfield slot.
- Standard lab setup: will have 6 slots, accommodating DAPI, GFP, RFP, and Cy5 while providing additional space for further upgrade.
- Automated research-grade microscopes could offer 8 to 10 slots for multi-color imaging and other specialized filters, like polarization/FRET filters
Does It Support Simultaneous DAPI, GFP, and RFP Imaging?
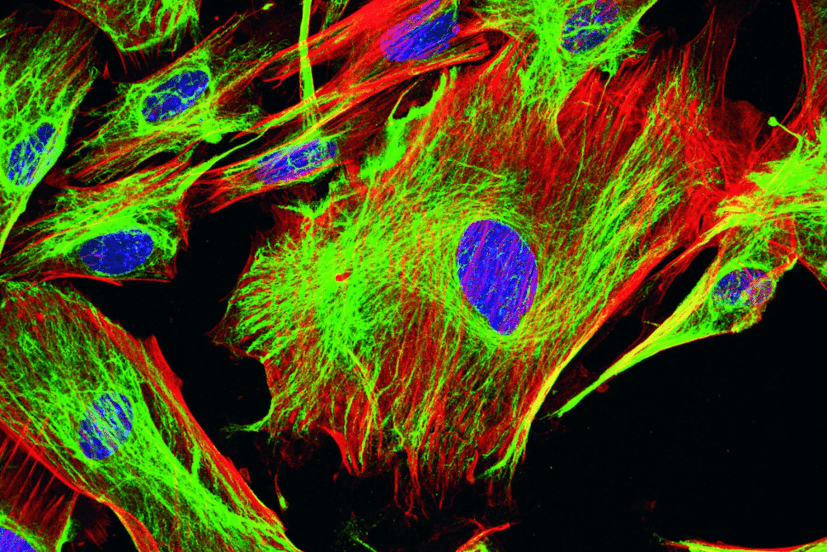
DAPI (blue), GFP/FITC (green), and RFP/mCherry (red) constitute the traditional “three color combination” in life science research. Being spectrally non-overlapping, this combination is fully compatible with standard fluorescence microscope systems (i.e., allows for concurrent staining of cell nuclei, target proteins labeled in green, and cytoskeletal components colored in red). However, the term “concurrent observation” may imply:
- Sequential Channel Imaging (Most Common): A DAPI, a GFP, and an RFP filter cube can be mounted on the revolver. The microscope will then rotate the revolver to capture images in each channel individually.
Pro: Pure signals with minimal crosstalk
Cons: Mechanical switching requires time (typically several hundred milliseconds). Not suitable for ultra-fast live-cell dynamic imaging
- True “single-view three-color” imaging (multi-band): if a triple-band filter is installed together with a broadband light source.
Result: Red, green, and blue fluorescence can be observed simultaneously through the eyepiece.
Trade-off: Background noise increases significantly, Difficult to quantitatively analyze individual channel intensity afterward
3. If I Want to Observe Far-Red Channels Such as Cy5/Cy7, Does the Light Source and Filter System Support It?
If you are working with Cy5 (647nm) and Cy7 (750nm+) fluorophores, far-red filter cubes must be present in your turret. Although a 6-position turret suffices in terms of physical space, whether or not you will be able to view these fluorophores hinges upon the optimization of your light source and filters for the NIR range.
Filters (The “Gatekeepers”)
- Cy5 (~650nm-670nm): Most current research microscopes will work well with Cy5 cubes, which can be readily added.
- Cy7 (~750nm+): This belongs firmly in the NIR range. Cy7 cubes are not included in standard filter sets and need to be specifically purchased and installed.
Light Source (The “Energy Source”)
Your light source strength at the red and infrared wavelengths is essential:
- Mercury Lamps: The spectral intensity declines sharply beyond 600nm. Although Cy5 may be used to a minimum extent, Cy7 imaging is impossible.
- Metal Halide Lamps: Provides higher energy in the red range compared to mercury, thus adequately supporting Cy5.
- LED light source: crucial! Only those systems having a specific LED module at the red or near-infrared wavelengths (e.g., 630nm and 740nm) are capable of Cy5/Cy7. If the light source only consists of UV, Blue, Green, and Yellow channels, then Cy5/Cy7 might be unsupported. Lasers (confocal): In confocal systems, a 633nm or 640nm laser is included for Cy5 imaging.
Detector (The “Sensor”)
A signal that the naked eye cannot see demands highly sensitive detection methods:
- Quantum Efficiency (QE): Traditional CMOS/CCD cameras exhibit a considerable decline in QE beyond 700nm.
- High-Quality NIR Imaging: For achieving Cy7 imaging, one will need to use either back-thinned sCMOS cameras or an IR-enhanced sensor.
4. Comprehensive Hardware Checklist
| Key Concern | Recommended Specification |
| Filter turret capacity | At least 6 positions reserved for DAPI, GFP, RFP, Cy5, Cy7, plus one spare slot |
| Cy7 support | Broadband LED or xenon light source; custom filters; NIR-enhanced camera |
| Three-color imaging | Sequential imaging with filter switching is recommended instead of triple-band filters (unless only used for rapid visual screening) |
| Objective quality | Preferably 60×/100× Plan Apo Oil objectives (NA 1.4) |
In case you discover that the signal for Cy5 is very faint, apart from verifying NA and QE, which you mentioned, I would also urge you to verify that the internal optical lenses of your microscope have near-infrared anti-reflection coatings. There are instances where some of the old glass parts of the microscope have an absorption rate for infrared light.