Embryo development happens fast. Cells divide, migrate, and reshape tissues in minutes or hours. Capturing those changes without damaging the sample is one of the biggest challenges in live imaging.
- Light Sheet Microscopy
- Confocal Microscopy
Both can generate beautiful 3D images. Both are widely used in research. But when it comes to tracking embryo development over time, they behave very differently.
Choosing between Light Sheet Fluorescence Microscopy (LSFM) and Confocal isn’t just a technical debate—it’s about whether your sample survives long enough to tell its story.
Let’s break down which one actually wins for embryo development tracking without the heavy textbook jargon.
The “Sunburn” Problem: Why Embryo Development Imaging Is So Demanding?
Embryos are arguably the most high-maintenance samples in the lab. They’re dynamic, 3D, and incredibly sensitive to light.
In many experiments, researchers want to image entire embryos continuously for hours or even days.
| Requirement | Why It Matters |
| Imaging speed | Developmental events can happen within minutes |
| Low phototoxicity | Embryos are sensitive to light exposure |
| 3D imaging | Most developmental processes are volumetric |
| Long-term stability | Experiments may run for 24–72 hours |
| High resolution | Cell-level tracking is often required |
Traditional widefield or older imaging setups often fail because they hit the embryo with too much energy.
Imagine trying to film a sleeping baby by blasting them with a stadium floodlight—they’re going to wake up (or in the embryo’s case, stop developing). This phototoxicity is the number one reason researchers lose weeks of work. To track development, you need a tool that’s fast enough to catch the action but gentle enough to keep the “baby” alive.
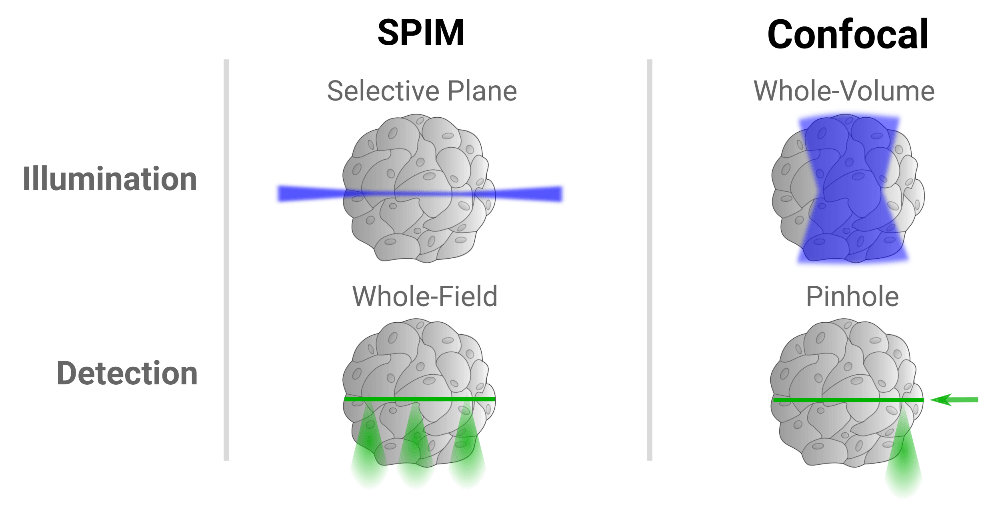
Light Sheet vs. Confocal Microscopy: Which one wins for embryo development tracking?
- Light sheet favors speed and sample health.
- Confocal favors resolution and precision.
| Feature | Light Sheet (LSFM) | Confocal (Point Scanning) |
| Survival Rate | Superior. Perfect for multi-day imaging. | Moderate to Low. Risk of “frying” the sample. |
| Speed | Blazing fast. Captures 3D volumes in seconds. | Slower. Point-by-point scanning takes time. |
| Resolution | Great, especially for large-scale tracking. | Top-tier. Better for sub-cellular details. |
| Setup Ease | Tricky. Requires special sample mounting (FEP tubes). | Easy. Put it on a slide or dish and go. |
| Data Size | Massive. Be ready for Terabytes of data. | Manageable. Standard file sizes. |
Light Sheet: The Gentle Giant
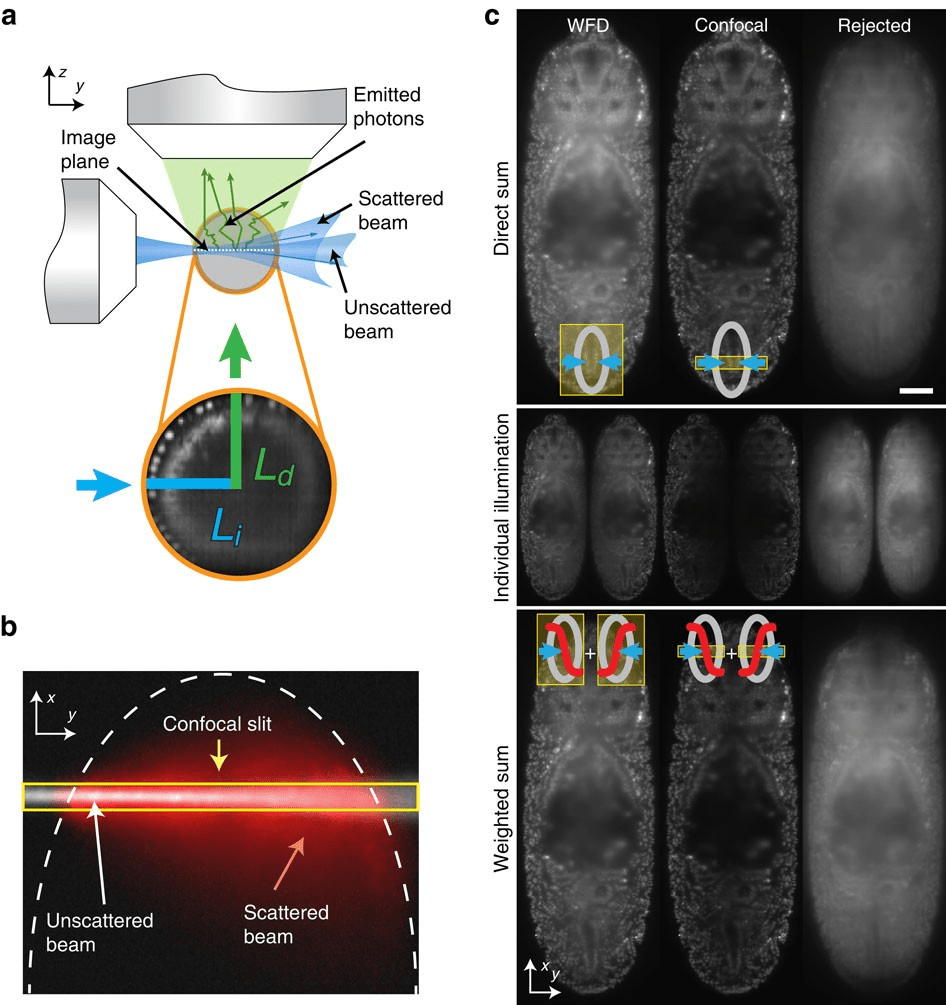
Think of Light Sheet as a precision deli slicer. Instead of bathing the whole embryo in laser light, it illuminates only a single thin “sheet” of the sample at a time.light sheet systems are widely used for whole-organism imaging, especially for embryos.researchers can image entire embryos while they develop.
This design has several advantages:
- extremely fast image acquisition
- minimal exposure of the sample to light
- efficient 3D imaging of large specimens
Because the camera captures the whole slice at once (rather than scanning point-by-point), it’s insanely fast. If you’re tracking a zebrafish heart beating or rapid cell migration in a fruit fly, Light Sheet is usually the gold standard. It keeps the embryo in a happy, healthy environment for days on end.
Confocal: The Reliable Detail-Seeker
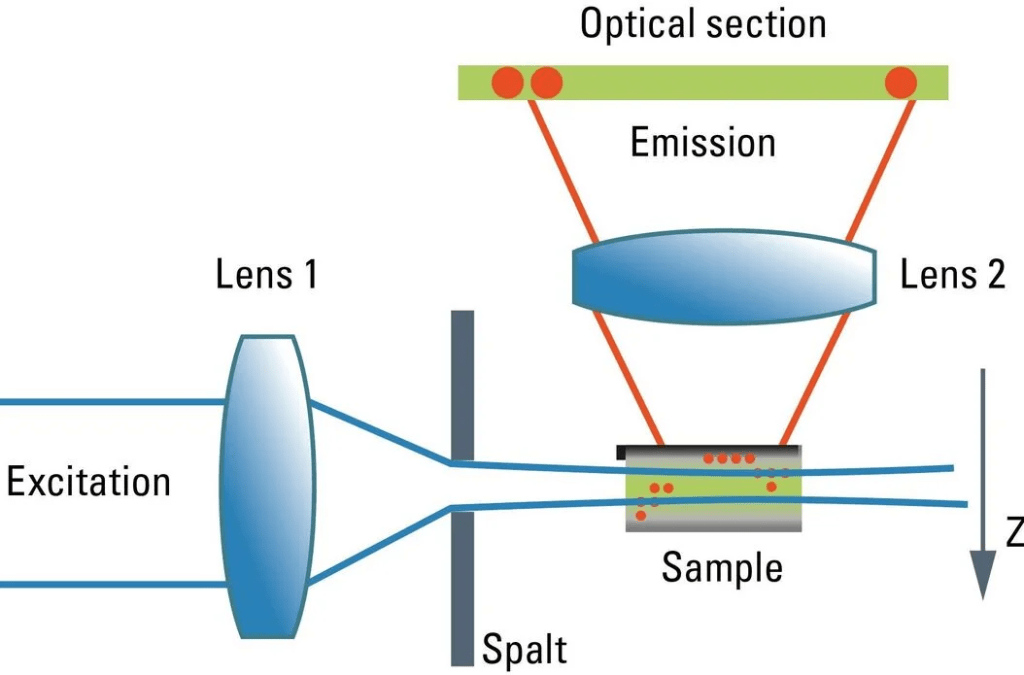
Confocal is the standard workhorse of the imaging world. It’s amazing for high-resolution snapshots and getting crisp images of thinner samples.A laser scans the sample point by point, and a pinhole blocks out-of-focus light. This produces sharp optical sections and excellent image contrast.
Confocal microscopes are famous for:
- strong optical sectioning
- high spatial resolution
- reliable fluorescence imaging
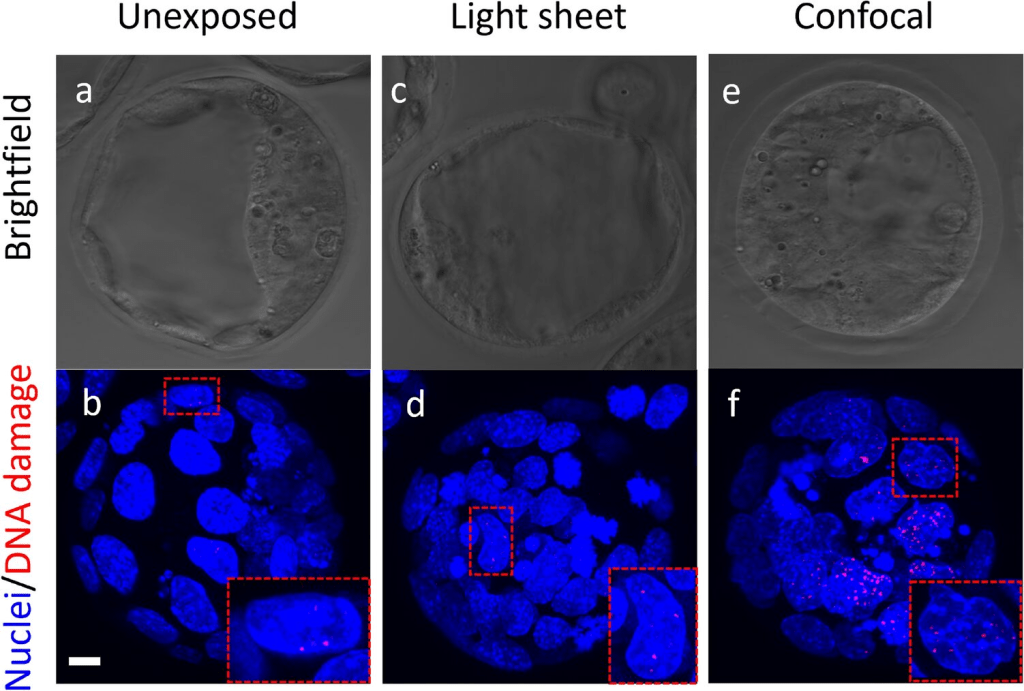
For smaller samples or detailed cellular studies, confocal imaging can produce extremely precise results.But the scanning approach also introduces some limitations when imaging large, dynamic samples like embryos.
“However, for thick embryos, Confocal has a “tax.” To get an image, it has to pass a laser through the entire depth of the tissue to hit one specific spot. This means even the parts you aren’t currently “filming” are getting blasted with radiation. For a 48-hour developmental study, this often leads to bleaching—where your fluorescence just disappears—or worse, developmental arrest.”
Limitations: Why Researchers Still Lose Sleep Over This Choice
No imaging system is perfect.Light sheet microscopes come with their own challenges.Even though Light Sheet sounds like the winner for live imaging, many people still hesitate.Why?
- First, there’s the mounting headache. You can’t just throw an embryo on a glass slide for a Light Sheet microscope; you often have to suspend it in a tube of agarose. It’s an art form in itself.
- Second, it’s the data mountain. Light Sheet produces so much data so fast that your lab’s hard drives will be screaming for mercy within a week. If you don’t have a massive server and a dedicated bioinformatician, Confocal might feel like the “safer” bet for shorter experiments.
Different microscopes perform better depending on the biological question.
| Research Scenario | Better Choice | Why |
| Whole embryo development tracking | Light Sheet | Fast volumetric imaging |
| Long-term live embryo imaging | Light Sheet | Low phototoxicity |
| Protein localization in cells | Confocal | Higher spatial resolution |
| Imaging subcellular structures | Confocal | Strong optical sectioning |
| Morphogenesis over time | Light Sheet | High temporal resolution |


